CBEMresults
MCBP Summer Water Quality Testing Campaign with Hackuarium
Hammerdirt, Biodesign for the Real World, AGiR! and the Hackuarium are doing community based environmental monitoring, specifically testing water quality for the Montreux Clean Beach Project around Montreux Bay in Lake Geneva.
The first 'qualification' day to make sure the protocols and facilities were set to go was run on 7 June 2016.
Qualification Day
Hammerdirt volunteers met with the technical advisor from AGiR! and held a water sampling and processing 'qualification' day (Q-Day) prior to the beginning of the Water Quality Testing (WQT) pilot project.
Objectives of Q-Day
- Combine a litter survey with the collection of water samples
- Collect samples in accordance with the protocol for the WQT project
- Prepare culture medium using available laboratory equipment
- Collect samples without introducing contaminants
- Prepare samples without introducing contaminants
- Execute all tasks following laboratory safety regulations
- Verify that all laboratory equipment is operational
Meeting objectives
The success of Q-Day was based on the response to the following questions:
- Were the samples collected and plated within a 6-hour window?
- Was the litter inventory completed prior to leaving the site?
- Did field team observe proper hygiene and sampling protocols?
- Did all laboratory equipment function? Were the team members able to use equipment?
- Did the negative controls show any signs of growth?
- Did the positive controls show growth in accordance with historical results?
The survey sites for Q-Day
- Positive control Vauchère river
- Negative control : tap water
- Unknown : Bay de Vidy
Combining a litter survey with water sampling
A comprehensive litter survey can take up to two hours (depending on the size of the beach). For the Q-day exercise hammerdirt chose a small beach (Vidy) not far from an urban center (Lausanne). Two risks were immediately identified:
- Cross contamination of samples from handling litter and water sampling
- Running out of time within a 6-hour window to plate the samples
It was decided that initially the litter would be collected at the site by all team members and then, at the discretion of the team members, one individual would cease handling litter and collect the required samples.
11:50 Litter collection and inventory
12:50 Sample collection
13:40 Arrival at the laboratory
Collect samples in accordance with the protocol
The location was easy to access. The team packed soap, hand sanitizer and disposable gloves specifically for water sampling operations. The designated individual ceased handling litter and washed hands with soap and water. Clean, disposable gloves were used for the sampling process. Samples were collected by wading into the water and plunging the sampling tube roughly 50cm deep. The sampling tube was uncapped underwater and filled completely. Once the sampling tube was out of the water a portion was poured off and the sampling tube was capped and put on ice. All team members washed hands and used hand sanitizer at the end of the operation.
Prepare culture medium using available laboratory equipment
For Q-Day operations we used a dehydrated culture media, eosin methylene blue agar or Levine media produced by Oxoid (product number CM0069). The culture media that was ordered and that will be used for the WQT project, ECA Check Easygel, had not arrived by the scheduled date for this exercise. Such media requires significantly more preparation and handling than the ECA Check Easygel. The following steps needed to be completed prior to plating (within the 6-hour limit):
- Suspend 37.5g in 1 litre of distilled water.
- Bring to a boil to dissolve completely.
- Sterilise by autoclaving at 121°C for 15 minutes.
- Cool to 60°C
- Shake the medium in order to oxidise the methylene blue
- Pour medium into sterile petri dishes
- Cool medium to solid and introduce water samples
- Prepare samples without introducing contaminants
Collected samples were stored in the laboratory specimen refrigerator at 4c while the chromogenic medium was prepared. The work surface, a fume hood, was sterilized with 70% ethanol prior to pouring the chromogenic media into disposable, sterile petri dishes. The surface was sterilized again prior to plating of the water samples.
Sterile, disposable pipettes were used and changed for each water sample. Participants used disposable latex gloves and a Bunsen burner was lit during all pouring and plating operations.
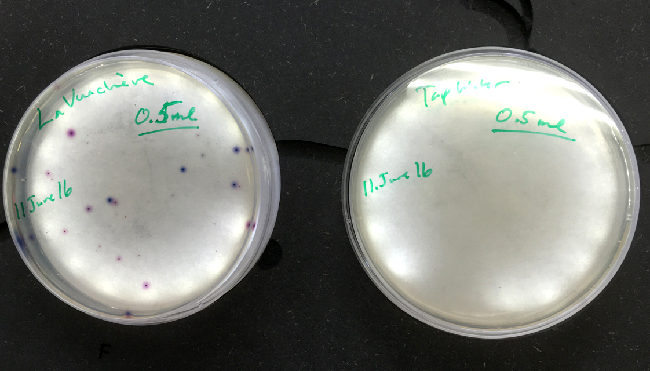
Three samples from the positive control and the unknown were plated as well as one sample from the negative control (tap water).
Plated samples were covered and placed in the incubator at 37oC for 24hours.
Were the objectives met?
- Were the samples collected and plated within a 6-hour window? Yes, samples were collected at 12:50 and plated at 18:30.
- Was the litter inventory completed prior to leaving the site? Yes the results can be seen here :Vidy inventory
- Did field team observe proper hygiene and sampling protocols? Yes, field participants prepared and utilized a field hygiene kit including soap, hand sanitizer and disposable gloves. The depth of water sample was observed.
- Did all laboratory equipment function? Were the team members able to use equipment? Yes, although prior organization and identification of the proper functioning equipment could have reduced the preparation time.
- Did the negative controls show any signs of growth? No, the negative controls exhibited no growth. Indicating that no contamination occurred during sample handling and processing.
- Did the positive controls show growth in accordance with historical results? Yes, in accordance with the experience of the technical advisor.
Remarks
- Field staff will need to organize activities appropriately especially in regards to the times required to inventory litter and the 6-hour time limit for water samples.
- A bottle of water should be included in the field hygiene kit to rinse hands.
- A dedicated space needs to be attributed in the lab for this project.
- The incubator needs to be reserved ahead of time for sample processing (avoid conflicts with other projects).
- The level of the fume hood should be verified, the cooled plates showed a slight unequal distribution.
- A training session with ECA Check Easygel should be scheduled.
Initial Conclusions
An opening day can be scheduled, hammerdirt staff are competent and all the necessary equipment is available at the lab.
Results of Microbiological Analyses
The samples were removed from the incubator after 24hours of incubation at 37°C. At least 4 colony morphologies were visible on the Levine media plates, including metallic green CFU, initially considered our bioindicator bacteria, E. coli (although 2020 - this is now being further investigated!) - and also various purple and pink colonies, and scored.
There were no visible colony forming units in the tap water or the sample taken from Vidy beach. The number colony forming units of E.coli counted from the Vauchère samples are presented in the table below.
Colony forming units counted: Vuachère (positive control, river)
| Counts | Vauchere 1 | Vauchere 2 | Vauchere 3 | Avg | Std Dev |
|---|---|---|---|---|---|
| AGiR! counts | 7 | 3 | 29 | 13 | 14 |
| Hammerdirt counts | 7 | 3 | 27 | 12 | 13 |
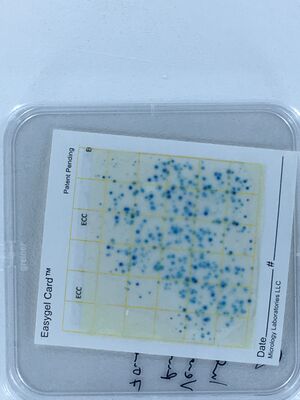
The ECA Check EasyGel material (pre-treated plates and individual liquid media bottles) arrived on June 11th. A control of 0.5ml of the same tap Water and Vauchère (saved in the refrigerator) samples were plated. With the EasyGel plates, it is much simpler to score the colony forming units than on the Levine media plates. Thank you, Hackuarium, for the microgrant!
Summer Sampling Results around Montreux: 8 weeks of sampling, two consecutive summers
For the Montreux Clean Beach Project collaboration between Hammerdirt and Hackuarium, water sampling at three sites around Montreux Bay over eight-week summer seasons was pursued, in parallel with shoreline litter surveys to quantitate trash abundance.
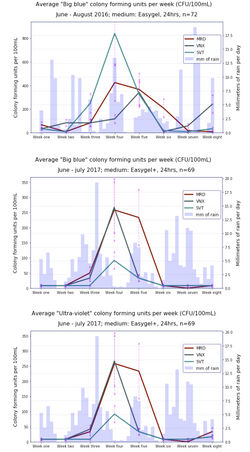
Please [see this] for an 'infographic' showing plates from an average sampling day (once a week for eight weeks each summer). We gathered a lot of good data, with our triplicate plate sampling protocol. (The negative control of tap water, and positive control of river water only were allowed one plate each week.) Clearly, our bioindicator for dirty sewage water getting into the bay, E. coli, showed up during both years sampling campaigns and peaked during the local music festival, the Montreux Jazz, when about 20000 more people come to the area each year. Other bacterial species, identified on the chromogenic plates used, also increased in abundance, but more data analyses could still be done.
We also tested out a new sort of card, in addition to using EasyGel plates in classic petri dishes, in 2017 (example shown on right). These could be less expensive, for shipping especially.
After our second summer of sampling, when we included an extra indicator that fluoresced to confirm the presence of E. coli in the Micrology Lab plates (ECAPlus, as shown to [here]), the peaks of water pollution observed around Montreux were still quite evident, even in spite of lower levels of rainfall, in comparison to the previous summer. While E. coli CFU were found to be at levels much higher than those desired for Swiss public waters, they were however below European standards.
(To note: for recreational waters, 100CFU/100ml are the Swiss limits, while the EU allows 500CFU/100ml.)
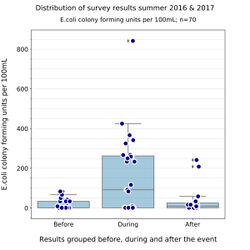
Bioindicator abundance is shown for the 2 summers of sampling
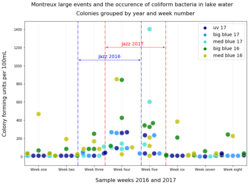
to the left, and a summary figure for the results of the two years of eight-week sampling periods can be found below, on the right.
As can be seen, pollution of Montreux Bay waters is clearly increased during and after the Jazz Festival. The box-plot distributions are another good way to visualise these results, below right.
The primary data repository and coding details are here.
We hope these data will not only influence decisions about renovations of the sewage treatment plant in Montreux (at Clarens), but also inspire others to pursue further 'clean beach' projects internationally.
To note: we have furthermore visited the local sewage treatment plant at Clarens (see below, right), and discussed these results widely, for instance, in medium.com and at meetings, like the EPFL professors' life sciences retreat. We still have plans to provide all data in the context of Zenodo (and perhaps a journal article, even) to encourage not only further analyses of more of the microbial data (which is about much more, again, than simply E.coli abundance), but also more participative research in this regard, internationally.
A NoJazz Summer for 2020
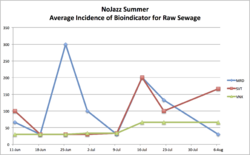
BIG NEWS: We've done more sampling during the Covid-19 Crisis, as the Jazz was cancelled. First sampling was done June 11, with plating on LB and Levine media, and low baselines were observed at all 3 sites, particularly of our bioindicator for raw sewage. Both ordinary LB and selective Levine media plates were used for inoculations. These sampling data are already collected, but it depended upon participation by interested people, in order to get the full 8 weeks done... (managed it in 9 weeks! ;) We were glad to do a local beach litter survey to complement the microbial work in the course of this NoJazz summer campaign, too!
- Median bioindicator levels on average as shown on Levine plates from the water sample that day were not super for a swimming beach: 700CFU/100mls (7-fold above the acceptable levels for Swiss recreational waters that day.)
Graphed results after this NoJazz summer of sampling showing the bioindicator for raw sewage (metallic green CFUs) are shown below:
To note: the 4th week we also did 3 plates for the positive control river samples, and 14,500CFU/100ml
of the bioindicator bacteria were found on average. Furthermore, the first couple of weeks' samples were taken after very rainy weather in preceding days.
Also to note: more data is already being assessed (with the second week at Clarens beach mentioned above and including some extra controls on shallow vs 1m deep sampling). See figure below for the full 8 weeks worth of data (taken over 9 weeks - everyone, even during a pandemic, should be able to have some time off... ;)
We even got some 16S sequence data for species ids! Apparently on the levine media, 'metallic green' colonies can even be Aeromonas but most are indeed wild E. coli! More of these data (à la 'microbiome analyses' to id species with universal rDNA primers like [in this old wiki page]) will be included in the academic article now under revision... This is all such a great experience, and we can attest that the procedures of peer review are clearly something that benefit a manuscript. (and us all? esp if can avoid the ones more like PR announcements... vive open science, but better comms mechs are really needed all around!)
In short, the NoJazz control showed: out of 24 sampling sites around Montreux Bay, 7 sampling sites did have scattered incidents of pollution with 100 CFU/100ml or more over the sampling period, but just 3 incidents over 200CFU/100ml water sample.
(100CFU/100ml is the local limit for recreational waters - but again, the Bay of Montreux is not monitored as a swimming beach)...
Seems different from our two years with the Jazz on. But the team's crunch of numbers via python coding will tell us more, also adding in the rain data (all thanks to Roger, Masoud and Carlos !) whew.
The time is ripe for improvements, and other neighborhoods' waterways and STEPs are getting updates, so who knows, particularly if this publication gives the region more international interest.
on va voir! et bientôt!
The article was accepted!
Data is ready, more than 7gigabytes archived in Zenodo. & as of spring 2022, these data were downloaded 50 times already! =)
Here is the github for analyses (you can launch 'mybinder' from there to play with it all, too).
We filled in the license contract and have a doi already, too:
Article: Participatory research to monitor lake water pollution
Journal: Ecological Solutions and Evidence
Article DOI: 10.1002/2688-8319.12094
To note also: the graphs will have a different look in the final paper! As always, the review process is enlightening! We were thankful for the input and the interesting time, although the amount of time... well, enough said for now... ;)
& More
Anyone interested in helping getting more responses to our note to the Montreux Commune and Jazz organisers? ;)
Getting a local team from Montreux to do the monitoring is the big hope during the summer season of 2022.
Avoir une équipe locale de Montreux à faire la surveillance durant la saison 2022 serait super...
The surprise is that Micrology Labs has been bought out and is now Incorporated, via a not entirely straightforward business transaction, it seems... Nonetheless, the former owner has kept the best - the more ecological R-Cards - as the key product in his new company, Roth Bioscience LLC, which we will most likely use for the summer of 2022.
Want to join in for fun in 2022? contact hello (at) hackuarium (point) ch
Here is the new page for the Campaign 2022 of the Montreux Clean Beach Project!
Here is the official invitation for the big launch of the MCBP 2022 (in French, bien sûr)!
Participez au lancement de notre campagne 2022 sur la qualité de l’eau du lac !
L'association Hackuarium organise une sêrie de mesures de la qualité de l'eau du Léman.
Comme lors des campagnes précédentes (2016, 2017 et 2020)*, nous effectuerons des tests microbiologiques mesurant un bio-indicateur important de la pollution des eaux usées non traitées, E. coli, qui fait partie de notre propre microbiome. La campagne de mesures s’étendra sur une période de 8 semaines, du lundi 13 juin au vendredi 5 août 2022, avec des échantillonnages supplémentaires pendant le festival de musique, le Montreux Jazz.
Vous êtes cordialement invité-e-s à participer au lancement de cette campagne de surveillance:
Lundi 13 juin 2022, à 16h30
à Montreux, à côté du Sauvetage (La Vigie) devant l’auditorium Stravinsky,
sur le quai de Vernex (https://goo.gl/maps/XUgRrcz6eTy4q3gW6).
La séance est publique.
Les volontaires d’ Hackuarium vous montreront les techniques de prélèvement des échantillons et la méthode d'analyse de la teneur en bactéries.
Si vous le souhaitez, vous pouvez également apporter votre propre échantillon d'eau dans une bouteille propre, pour effectuer un test.
(Tests limitées à 12 participants et résultats connus après 48 h d'incubation)
Rejoignez-nous dans notre campagne de la recherche participative !
Si vous avez des questions ou si vous voulez participer à la campagne complète, écrivez-nous svp à [[1]].
*article publié en 2021: https://t.co/IaoP9SxJdh
- translation help always in demand =)
To go back to the initial project page