BioP Colour Bacteria
Microbiology of pigment-producing bacteria
Contribution to Open Science School’s Co-lab Biomaterial EPFL & Hackuarium, December 2016, by Vanessa Lorenzo Toquero Hybridoa
Introduction
- Growth conditions of microorganisms
- Physical (temperature, light), chemical (pH, osmolarity), toxicity (antibiotic, heavy metal), or nutrient requirement (auxotrophy, bioavailability) conditions.
- Pigments and physical nature of color.
Bacterial growth
Bacterial growth is affected by (1) temperature, (2) nutrient availability, (3) water supply, (4) oxygen supply, and (5) acidity of the medium.
Temperature
Theoretically, bacteria can grow at all temperatures between the freezing point of water and the temperature at which protein or protoplasm coagulates. Somewhere between these maximum and minimum points lies the optimum temperature at which the bacteria grow best. Temperatures below the minimum stop bacterial growth but do not kill the organism. However, if the temperature is raised above the maximum, bacteria are soon killed. Most cells die after exposure to heat treatments in the order of 70°C for 15 seconds, although spore-forming organisms require more severe heat treatment, e.g. live steam at 120°C for 30 minutes. Bacteria can be classified according to temperature preference: Psychrophilic bacteria grow at temperatures below 16°C, mesophilic bacteria grow best at temperatures between 16 and 40°C, and thermophilic bacteria grow best at temperatures above 40°C.
Nutrients
Bacteria need nutrients for their growth and some need more nutrients than others. Lactobacilli live in milk and have lost their ability to synthesise many compounds, while Pseudomonas can synthesise nutrients from very basic ingredients. Bacteria normally feed on organic matter; as well as material for cell formation organic matter also contains the necessary energy. Such matter must be soluble in water and of low molecular weight to be able to pass through the cell membrane. Bacteria therefore need water to transport nutrients into the cell. If the nutrient material is not sufficiently broken down, the microorganism can produce exoenzymes which split the nutrients into smaller, simpler components so they can enter the cell. Inside the cell the nutrients are broken down further by other enzymes, releasing energy which is used by the cell.
Water
Bacteria cannot grow without water. Many bacteria are quickly killed by dry conditions whereas others can tolerate dry conditions for months; bacterial spores can survive dry conditions for years. Water activity (AW) is used as an indicator of the availability of water for bacterial growth. Distilled water has an AW of 1. Addition of solute, e.g. salt, reduces the availability of water to the cell and the AW drops; at AW less than 0.8 cell growth is reduced. Cells that can grow at low AW are called osmophiles.
Oxygen
Animals require oxygen to survive but bacteria differ in their requirements for, and in their ability to utilise, oxygen. Bacteria that need oxygen for growth are called aerobic. Oxygen is toxic to some bacteria and these are called anaerobic. Anaerobic organisms are responsible for both beneficial reactions, such as methane production in biogas plants, and spoilage in canned foods and cheeses. Some bacteria can live either with or without oxygen and are known as facultative anaerobic bacteria.
Acidity
The acidity of a nutrient substrate is most simply expressed as its pH value. Sensitivity to pH varies from one species of bacteria to another. The terms pH optimum and pH maximum are used. Most bacteria prefer a growth environment with a pH of about 7, i.e. neutrality. Bacteria that can tolerate low pH are called aciduric. Lactic acid bacteria in milk produce acid and continue to do so until the pH of the milk falls to below 4.6, at which point they gradually die off. In canning citrus fruits, mild heat treatments are sufficient because the low pH of the fruit inhibits the growth of most bacteria. (adapted from International Livestock Research Institute)
Strains
In this experiment, we will manipulate and grow different pigment-producing bacteria and observe their growth. Pigmentation is a characteristic that is common to many species of Bacteria. Pigments are light-absorbing compounds that are responsible for the colors that organisms display. Diverse groups of pigments are produced by organisms of the Bacteria domain, and they play important roles in the survival of the organisms which produce them. For example, the pigment xanthomonadin protects the Bacteria Xanthomonas oryzae from damage due to light, also called photodamage (extracted from MicrobeWiki).
We will use 3 different strains in our experiment:
Micrococcus luteus
Gram-positive, Coccus, saprotrophic bacterium that belongs to the family Micrococcaceae. An obligate aerobe, M. luteus is found in soil, dust, water and air, and as part of the normal flora of the mammalian skin. The bacterium also colonizes the human mouth, mucosae, oropharynx and upper respiratory tract.
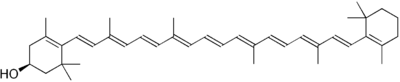
Chemical structure of xanthophyll, a yellow pigment found in Micrococcus luteus.
Micrococcus roseu
Gram positive bacterial cell that grows in the tetrad arrangement. The normal habitat for this Micrococcus species is skin, soil, and water. It derives its name from the pink carotenoid pigment that it secretes.

Chemical structure of canthaxanthin, a pink pigment found in Micrococcus roseus.
Janthinobacterium lividum
Aerobic, gram-negative, soil-dwelling bacterium that has a distinctive dark-violet (almost black) color due to violacein production. Its anti-fungal properties are of particular interest since J. lividum is found on the skin of certain amphibians, including the red-backed salamander (Plethodon cinereus), where it prevents infection by the devastating chytrid fungus (Batrachochytrium dendrobatidis).
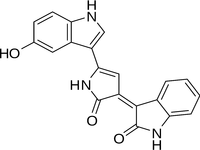
Chemical structure of violacein, a violet pigment found in Janthinobacterium lividum.
Materials
- Nutrient agar plates
- Luria Broth plates
- Luria Broth plates with Ampicillin (100 mg/ml), IPTG (1 mM) and tryptophan (2 g/l)
- Lb liquide
- Lb liquide + 1%Glycerol
Protocol: make your own experiment
Now you are free to draw colorful painting on your petri dishes, or experiment and use the bacteria on different conditions to check their growth. Please note the results and anticipate the results beforehand as an exercise. What is so special about natural pigments? Research ideas.
Pigments are normally produced by bacteria, plants, or even animals as a reaction to environmental stress or defense.
J. lividum
For example, the pigment violacein from J. lividum inhibits the toxic effect and growth of a fungus known as Batrachochytrium. This fungus causes a disease known as Chytridiomycosis in amphibians The disease devastated amphibian populations around the world, in a global decline towards multiple extinctions, part of the Holocene extinction. Because of this, understanding the uses of this bacteria has been of major interest. A study conducted in 2009 explored the effects of Bd and the use of J. lividium in the lab for survival. They used three experimental treatments: frogs infected with Bd, frogs given the bacteria J. lividium and frogs with the given bacteria and then exposed to Bd. Nearly all of the frogs exposed to Bd experienced mortality while none of the other treatments had any deaths. This effectively introduced the use of J. lividium as a possible method for Bd prevention in the lab setting.
Care
As you see, this bacteria can help you fight some diseases on amphibians (salamanders, frogs, etc). Lb agar plate, and a cozy 25 - 30 °C Temperature would make them happy. Purple babies will colonize your plate after 4 - 7 days. Wanna ink? The best is to transport them with a sterile spatula to a Lb (liquid) or Lb +1% glycerol (liquid) bottle, shake it constantly at around 270 rpm and a cozy 25 - 30 °C Temperature to make them happy.
- Note: They grow also in Ty and Ty + Gly is a boost for violacein pigment.
Micrococcus Luteus
Yellow mates that can be found almost everywhere: in soil, dust, water, air, in our skin… they are strong and they can live (almost) forever. Does a time machine of 30.000 years tell you something? Also, they block UV radiation… let’s go to the beach!
Care
Lb agar plate, and a warm up of 37°C Temperature for about 3 - 4 days would make them happy. Wanna ink? The best is to transport them with a sterile spatula to a Lb (liquid) shake it constantly at around 270 rpm at 37°C to have their shiniest bright yellow.
Micrococcus Roseus
Siblings of Luteus and living on mammalian skin, so, your skin as well! Uv protector and beautiful color pigment micro - factories. Are you thinking about a new tattoo?
Care
Lb agar plate, and a warm up of 25 - 37°C (they are chill and relaxed about the temperature) for about 3 - 4 days would make them happy. Wanna ink? The best is to transport them with a sterile spatula to a Lb (liquid). Shake them constantly at around 270 rpm at 37°C to have their beautiful and elegant pink-salmon babies.
Activities
- 1. Observe the different microorganisms
- 2. Play with the bacteria printer
- 3. Experiment different supports for their growth.
Experiments
Overall explanation of 1, 2, 3 steps. In groups of 3 people, choose 2 steps and 10 min experiment for 1st and 2nd step, 5min for 3rd step.
- 10 Kits * Experiment.
- Groups of 3
- Safety instructions and safety materials (safety about alcohol, gloves)
Kit 1
- 1 Mixed bacteria plate
- 1 Spatula
- 1 Lb agar plate
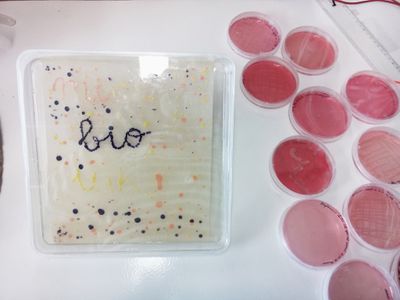
- Spot and isolate your colorful babies!
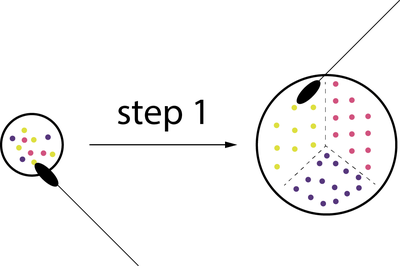
Experiment 1 description
- put gloves
- Clean your spatula
- open the mixed color petri
- take a sample of one of the colors at a time
- Scatter them into one area of the petri dish
- Clean your spatula
Kit 2
- 3 ink tonner
- 1 Paper
- 1 Brush
- Paint!
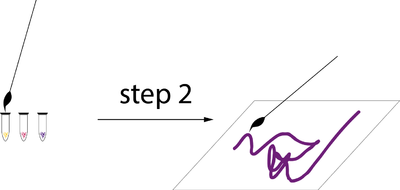
Experiment 2 description
- put gloves
- Clean your spatula
- take a sample of one of the colors at a time
- open the bacterial ink plastic container
- Soak your brush in the bacterial ink
- Paint your paper.
- Clean your spatula
Kit 3
- 1 Squared petri
- 1 Bio-printing session
- Make your digital design come alive!
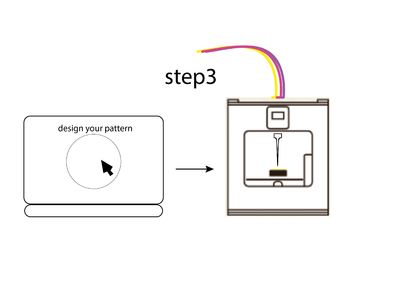
Experiment 3 description
- design your path on the computer
- print with it
General Material
- Heating plate/mixer
- Gas bottle (clean air circulation)
- Ethanol (Alcohol 96%)
- Gloves
- Brush
- Petri dishes
- Plastic containers
- Paper
- Kitchen paper
Biosafety
All the samples will stay at the lab and destroyed after 5 days (wil kept to document development), only the prototypes sprayed with ethanol or bleach will be kept.